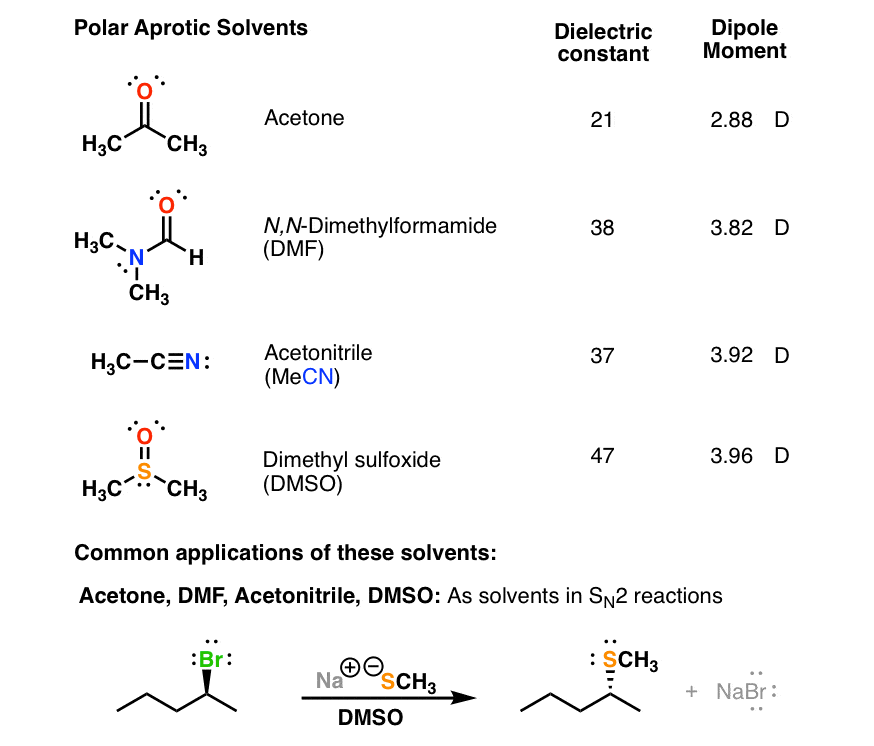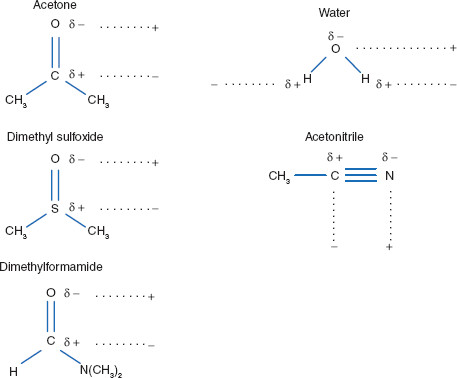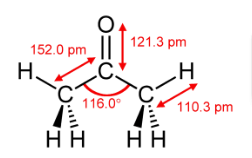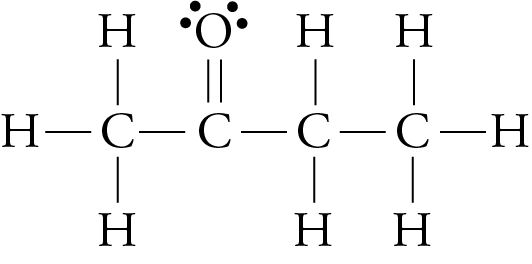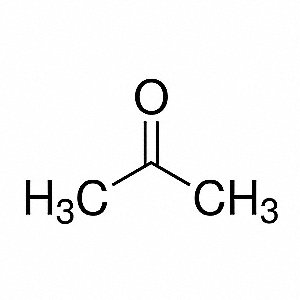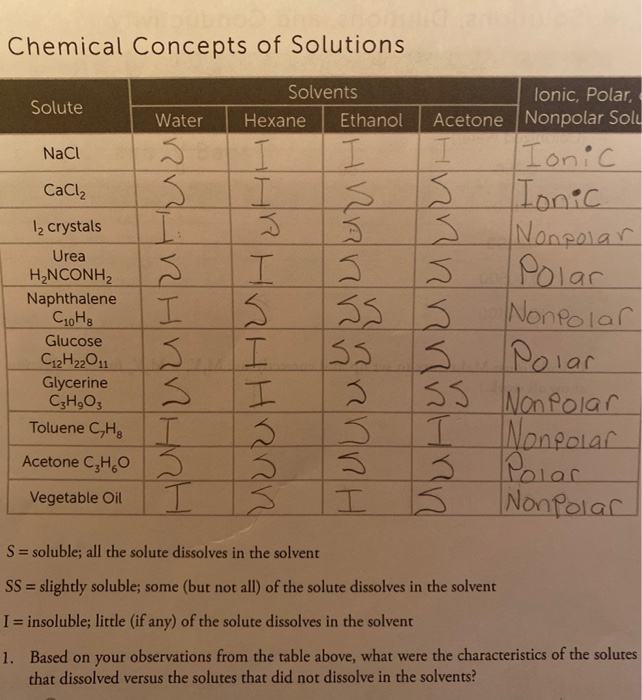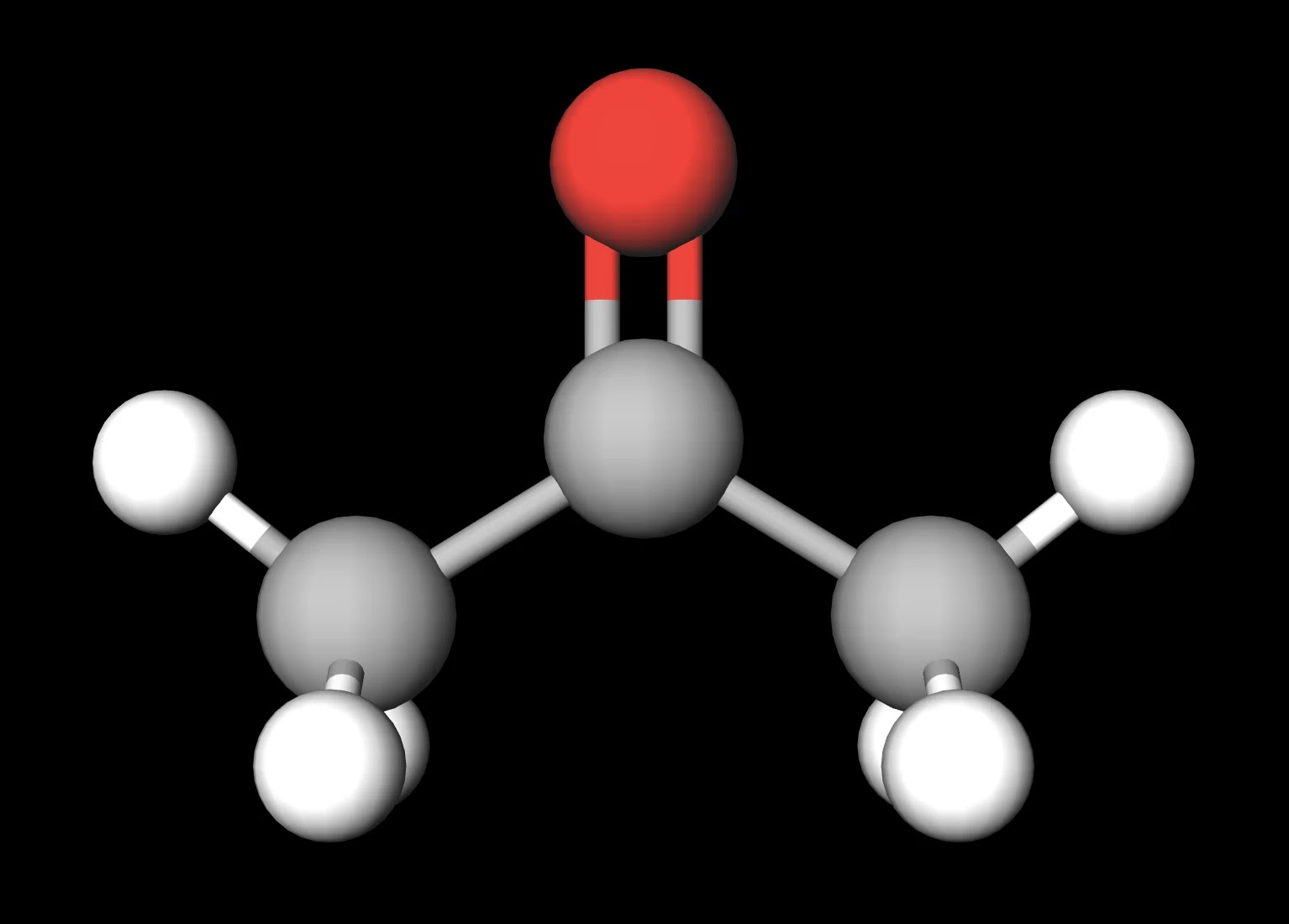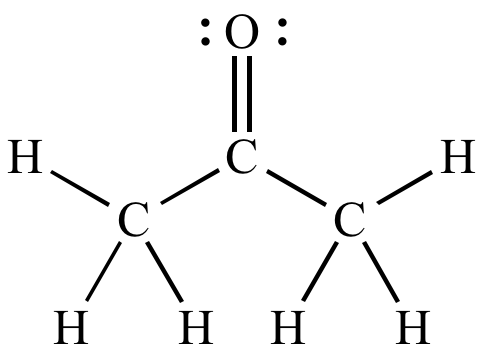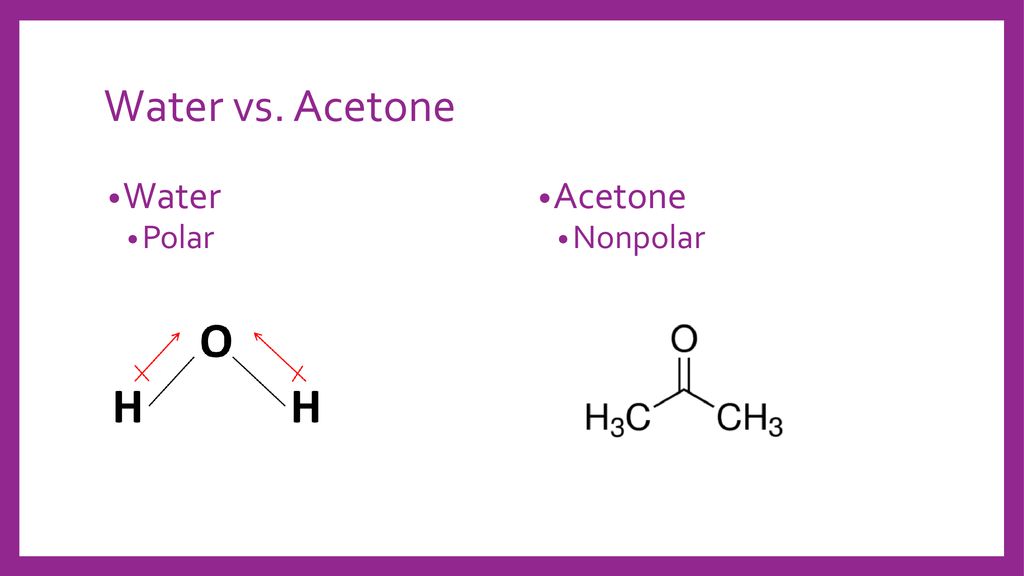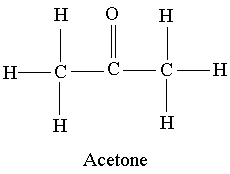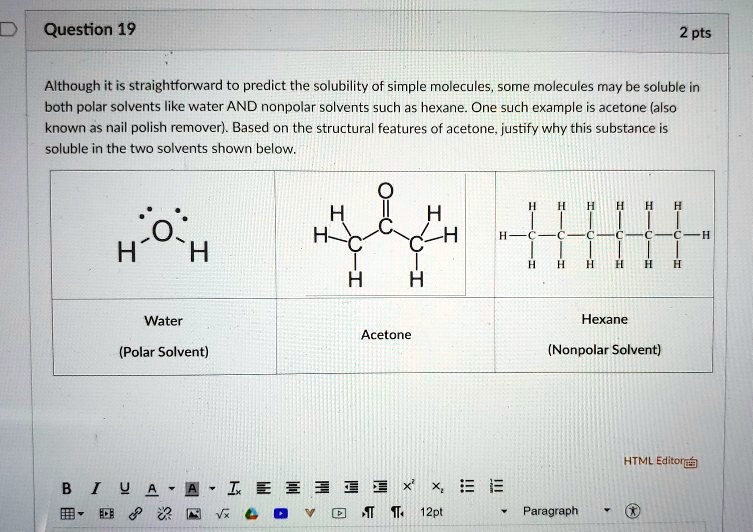
SOLVED: Question 19 2 pts Although it is straightforward to predict the solubility of simple molecules some molecules may be soluble in both polar solvents like water AND nonpolar solvents such as
What makes acetone a really good solvent? What allows it to dissolve both polar and non-polar molecules? - Quora
filosoffen.dk - what is metformin 500 mg used for | Is acetone polar or nonpolar molecule are mistaken
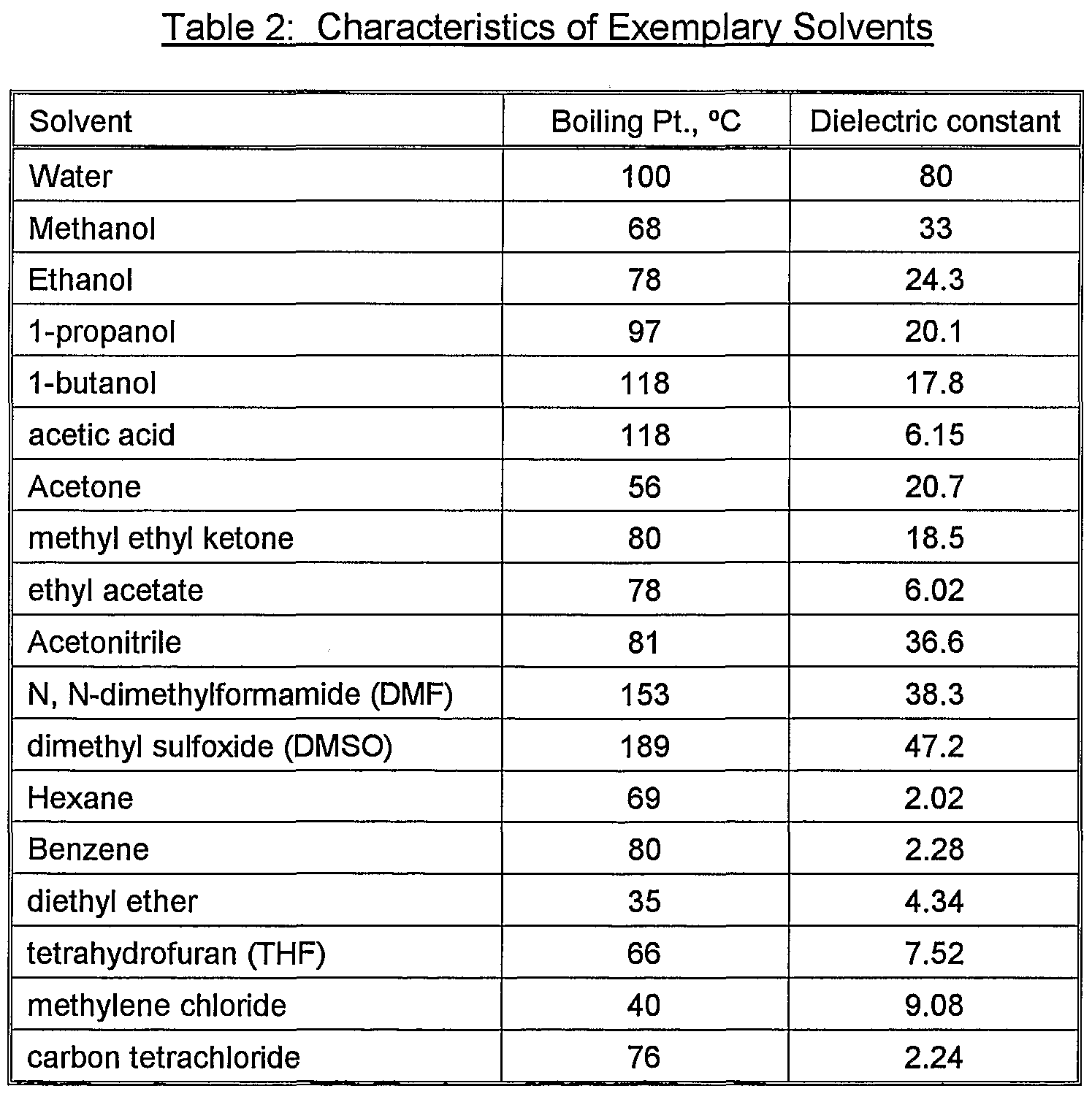
![PDF] Measurement and Modeling of Carbon Dioxide Solubility in Polar and Nonpolar Solvent | Semantic Scholar PDF] Measurement and Modeling of Carbon Dioxide Solubility in Polar and Nonpolar Solvent | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/4e1fdbc8b08394d57fd8a65ca57454ec1bcfb94d/2-Table1-1.png)