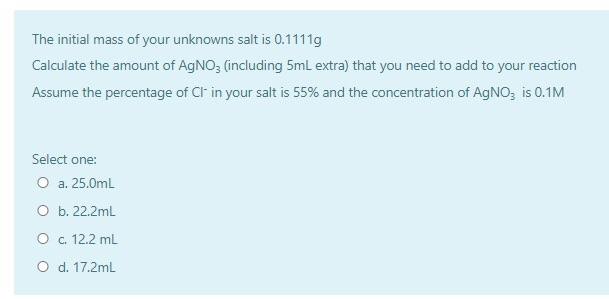
SOLVED: Calculate the approximate volume (in mL) of 0.1 M silver nitrate ( AgNO3) that will be needed to precipitate the chloride from a 0.1156 g chloride salt sample that is 55% weight

science chemistry dissolution reaction silver nitrate potassium chloride | Fundamental Photographs - The Art of Science
✓ Solved: Valence, Bonding, and Lewis Structures When a solution of salt (sodium chloride) in water is...
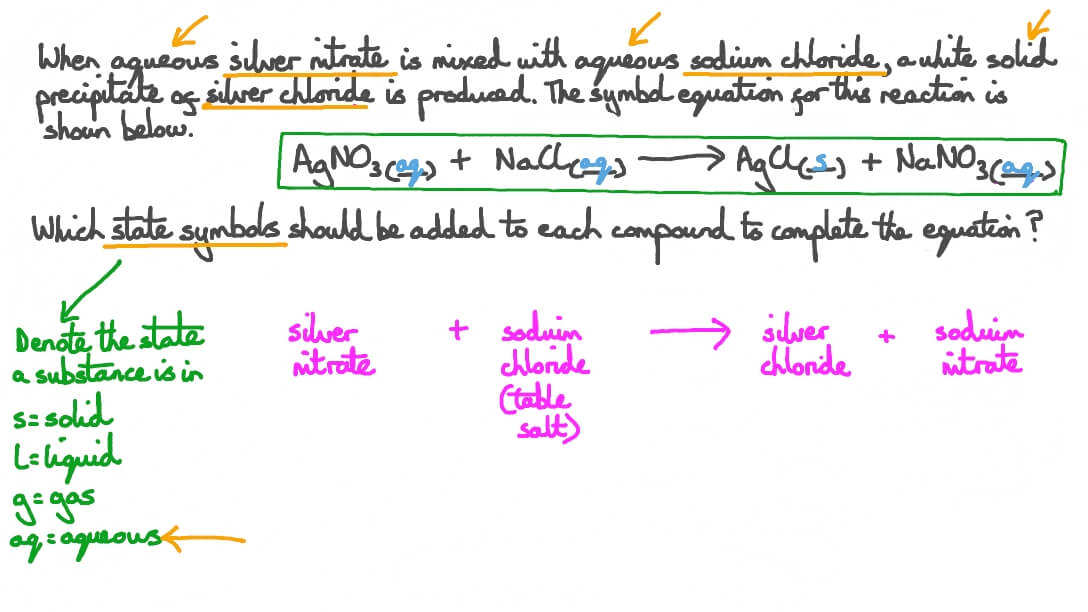
Question Video: Symbol Equation for the Reaction of the Precipitation of Agno3 with State Symbols | Nagwa